New mouse models with hypomorphic SUMF1 variants mimic attenuated forms of multiple sulfatase deficiency
Authors: Nicolina Cristina Sorrentino, Maximiliano Presa, Sergio Attanasio, Vincenzo Cacace, Martina Sofia, Aamir Zuberi, Jennifer Ryan, Somdatta Ray, Igor Petkovic, Karthikeyan Radhakrishnan, Lars Schlotawa, Andrea Ballabio, Cathleen Lutz, Nicola Brunetti-Pierri
Year: 2022
Sources: Journal of Inherited Metabolic Disease
Abstract:
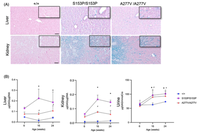
Multiple sulfatase deficiency (MSD) is an ultrarare lysosomal storage disorder due to deficiency of all known sulfatases. MSD is caused by mutations in the Sulfatase Modifying Factor 1 (SUMF1) gene encoding the enzyme responsible for the post-translational modification and activation of all sulfatases. Most MSD patients carry hypomorph SUMF1 variants resulting in variable degrees of residual sulfatase activities. In contrast, Sumf1 null mice with complete deficiency in all sulfatase enzyme activities, have very short lifespan with significant pre-wean lethality, owing to a challenging preclinical model. To overcome this limitation, we genetically engineered and characterized in mice two commonly identified patient-based SUMF1 pathogenic variants, namely p.Ser153Pro and p.Ala277Val. These pathogenic missense variants correspond to variants detected in patients with attenuated MSD presenting with partial-enzyme deficiency and relatively less severe disease. These novel MSD mouse models have a longer lifespan and show biochemical and pathological abnormalities observed in humans. In conclusion, mice harboring the p.Ser153Pro or the p.Ala277Val variant mimic the attenuated MSD and are attractive preclinical models for investigation of pathogenesis and treatments for MSD.
Category: journals